The Safety Syringe Global Market Report 2024 by The Business Research Company provides market overview across 60+ geographies in the seven regions – Asia-Pacific, Western Europe, Eastern Europe, North America, South America, the Middle East, and Africa, encompassing 27 major global industries. The report presents a comprehensive analysis over a ten-year historic period (2010-2021) and extends its insights into a ten-year forecast period (2023-2033).
Learn More On The Safety Syringe Market:
https://www.thebusinessresearchcompany.com/report/safety-syringe-global-market-report
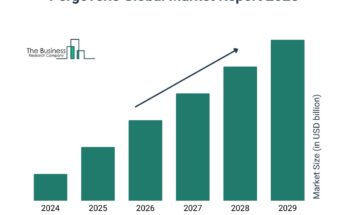
According to The Business Research Company’s Safety Syringe Global Market Report 2024, The safety syringe market size is expected to see strong growth in the next few years. It will grow to $12.6 billion in 2028 at a compound annual growth rate (CAGR) of 8.9%. The growth in the forecast period can be attributed to vaccination initiatives, rising geriatric population, increased focus on preventive healthcare, expanding biopharmaceutical industry, advancements in material sciences. Major trends in the forecast period include rapid technological innovations, emphasis on sustainable solutions, telehealth integration, customized solutions for specialized therapeutics, enhanced emergency response and preparedness.
The rising number of blood-borne diseases is expected to propel the growth of the safety syringe market going forward. A blood-borne disease is an infectious disease caused by microorganisms, such as bacteria, viruses, or parasites, that are transmitted through the blood. The use of safety syringes is a proactive measure to protect healthcare workers and patients from the risks associated with blood-borne diseases by reducing the likelihood of accidental exposures and promoting best practices in medical procedures. For instance, in September 2023, according to Contagion Live, a US-based news source, the blood-borne infection rates increased by 14% in 2020–2021, and there were 5023 confirmed cases of hepatitis C virus (HCV), with an estimated 69,800 infections. Further, around 43 states in the US also reported 107,540 newly diagnosed cases of HCV. Therefore, the rising number of blood-borne diseases is driving the growth of the safety syringe market.
Get A Free Sample Of The Report (Includes Graphs And Tables):
https://www.thebusinessresearchcompany.com/sample.aspx?id=13924&type=smp
The safety syringe market covered in this report is segmented –
1) By Product: Retractable Safety Syringes, Non Retractable Safety Syringes
2) By Therapy: Insulin, Glucagon-like peptide-I (CLP-I), Tuberculosis, Growth Hormones
3) By Application: Subcutaneous, Intramuscular
4) By End-user: Hospitals, Ambulatory Surgical Centers, Long-term Care Facilities, Specialty Clinics, Other End Users
Major companies operating in the safety syringe market are developing new products, such as passive needle guards, to gain a competitive edge in the market. A passive needle guard is a safety feature integrated into a medical device, such as a syringe, to enhance safety during and after use. For instance, in November 2021, Becton, Dickinson, and Company, a US-based medical device company, launched the BD UltraSafe Plus 2.25 mL Passive Needle Guard, an innovative addition to its drug delivery systems. This new solution extends the capabilities of the BD UltraSafe Plus needle guard platform, allowing the subcutaneous delivery of biologic solutions with volumes up to 2 mL and viscosities up to 30 cP. The design aims to address challenges related to the increasing viscosity and injection volume of biologics, providing a patient-controlled injection option for complex, high-viscosity drugs. The BD UltraSafe Plus 2.25 mL is designed to enhance the self-injection experience for patients, caregivers, and healthcare providers. The system aligns with the BD Neopak 2.25 mL Glass Prefillable Syringe, providing support for bio-pharmaceutical drug launches with convenience and ease of use.
The safety syringe market report table of contents includes:
- Executive Summary
- Safety Syringe Market Characteristics
- Safety Syringe Market Trends And Strategies
- Safety Syringe Market – Macro Economic Scenario
- Global Safety Syringe Market Size and Growth
.
.
.
- Global Safety Syringe Market Competitive Benchmarking
- Global Safety Syringe Market Competitive Dashboard
- Key Mergers And Acquisitions In Safety Syringe Market
- Safety Syringe Market Future Outlook and Potential Analysis
- Appendix
Top Major Players :
- Cardinal Health Inc.
- Retractable Technologies Inc.
- Thermo Fisher Scientific Inc.
- Medtronic plc
- Novo Nordisk A/s
Contact Us:
The Business Research Company
Europe: +44 207 1930 708
Asia: +91 88972 63534
Americas: +1 315 623 0293
Email: [email protected]
Follow Us On:
LinkedIn: https://in.linkedin.com/company/the-business-research-company
Twitter: https://twitter.com/tbrc_info
Facebook: https://www.facebook.com/TheBusinessResearchCompany
YouTube: https://www.youtube.com/channel/UC24_fI0rV8cR5DxlCpgmyFQ
Blog: https://blog.tbrc.info/
Healthcare Blog: https://healthcareresearchreports.com/
Global Market Model: https://www.thebusinessresearchcompany.com/global-market-model